In his TED talk on the UCLA campus in July of 2014, Reeve Foundation board member and T9-L1 para Mark Pollock sat tall and strong as he explained to a rapt audience what it is like to be blind, paralyzed, and operating in the “domain of the unpredictable.” This is his world, he said, because no one knows where the search for recovery from SCI will ultimately lead. The most promising and practical therapy, he believes, will likely be “a cocktail of cures.” And the search for that elusive technology is why he came from his home in Northern Ireland to UCLA in the first place.
Pollock, a medalist in rowing competition in the Commonwealth Games and a blind adventure athlete with several “firsts” to his credit prior to becoming paralyzed from a fall in 2010, came with his fiancée, Simone, to meet with Reggie Edgerton, the world’s leading researcher in the field of SCI and neuromodulation (more on this later). Edgerton’s many decades of clinical trials laid the groundwork for a relatively recent epidural stimulation trial at Fraser Rehabilitation Hospital in Louisville, Ky., led by Edgerton’s protégé, Susan Harkema. The trial made big waves, establishing proof of recovery of muscle function in complete chronic SCI [“Rob Summers: From Quad to Para and Beyond?,” Sept. 2011; “Cure Research: Looking to the Future,” Mar. 2013] and led to the Reeve Foundation’s latest research/fundraising initiative, “The Big Idea,” [Dec. 2014].
While The Big Idea seeks to gather more data by expanding the first epi-stim trial to upward of 30 subjects, Edgerton and his colleagues are creating smaller related trials, but with innovative twists: “Right now in one trial we are focusing on hand function specifically, but arm movement in general, on quads with levels below C4-5,” he says. “We are not focusing on ventilation, but that will come later.”
These new trials taking place at UCLA are part of a five-year NIH grant in its second year. “We’re trying to find what are the most optimal sites for stimulation,” says Edgerton, “what kinds of improvements they bring and what we are going to be able to achieve.” The upper limb trial involves six subjects, and a lower limb trial also involves six subjects. Their main purpose is to test a new stimulation technique that delivers electrical impulses transcutaneously to the spinal cord rather than via an implanted array of electrodes. The obvious advantage to this new delivery system is its potentially broad application, its timely feasibility, and improved cost. Edgerton says with sufficient funding he thinks a transcutaneous system could be ready for marketing in two years [see below].
Another new twist to these small trials is the testing of monoaminergic agents (drugs that work with receptor sites to improve nerve impulse transmission). “These drugs are about more than excitability of the cord. They must be selective to specific neuronal networks, and they must involve some kind of combination of turning on both inhibition and excitation in search of a balance,” says Edgerton. “You have a narrow window. If you get too much excitation, it will not get good results.”
The trials are small by design. “Smaller trials allow us to concentrate on individual subjects. I am convinced this is the best way to go,” says Edgerton. “When we do a large trial with hundreds and a generalized approach, we learn almost nothing new. But when we do small trials focused on individuals, we learn much more. It has been a real eye-opener. We kind of thought this might be the case, so that is why we did only five initially in the Louisville trial. We have learned enough now that we need to study more subjects, but we need to study comprehensively from each patient.”
A Trial of One
The most novel of trials is the one involving Pollock, who traveled from his home in Dublin to UCLA with his own Ekso Bionics robotic system, the first purchased by an individual, which he obtained in 2012. “My Ekso transmits all data back to a lab in San Francisco — number of steps, time, all the data,” says Pollock. “Using it from 2012 to 2014, psychologically I felt better standing and walking and felt physically better. But I couldn’t say there was any neurological recovery.” Edgerton likes this exoskeleton because it allows the user to take over some or all of the workload with his or her own muscle power. The hope is that adding stimulation will facilitate incremental recovery.
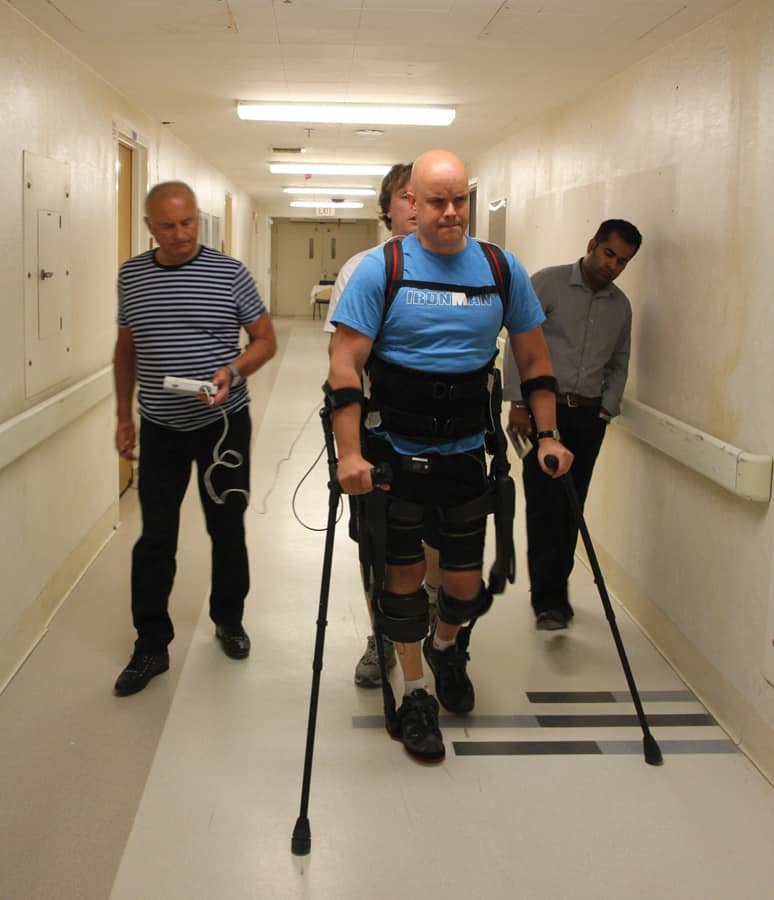
From February through May 2014, Pollock participated in a number of tests — walking in his Ekso and gathering data; then adding transcutaneous stimulation while walking in his Ekso and gathering more data; then taking the monoaminergic drug while Ekso-walking; and finally, combining all three modalities.
“Here is what was interesting for me,” says Pollock. “I had already done 250,000 steps in my robot when I got there, so I had a deep understanding of how it felt without stimulation. I was able to feel it was different with the stimulator on my back, and different with the drug, and I could certainly feel it was different with all three, in terms of both the sensation and my physical ability to move my legs. Trying each one separately and then combining all three, we got baseline data from which to build upon and explore different techniques, settings, and parameters.”
What were the specific one-man trial results? We will have to wait for published findings in a scientific journal. “We are working on the first draft of a specific manuscript on Mark right now,” says Edgerton. “It will probably be mid-year before it is accepted.”
The publicity surrounding The Big Idea, and now Edgerton’s new trials, inevitably leads mainstream media to spin advances as the “search for a cure,” but both Harkema and Edgerton tend to focus on improved quality of life as the immediate outcome. As for Pollock’s multi-modalilty trial and where it may lead, Edgerton says: “I think it would be more exciting and rewarding to walk around in one’s own environment rather than a rehab lab. It could have quality of life advantages. But if the robotic device is designed properly, it allows the subject to participate and be incrementally programmable. The ideal robotic device would work with complete paralysis, but the user could gradually train and regain more and more self-propelled movement. We hope to show this in future studies.”
“Training and regaining” is tied to the concept of neuromodulation. “We have pretty good evidence that neuruomodulation changes the physiological properties of movement we want to improve,” says Edgerton. “The neuromodulatory procedure [adding stimulation to activity such as stepping or grasping] changes properties of the neuronal system, and this enhances the system’s ability to perform. The circuitry of the cord must be neuromodulated, or re-educated, in order to improve the practical response.” That is the scientific way of saying the spinal cord has intelligence and can remodel itself.
Pollock wants to see all forms of stimulation developed, not only for what it means to him, but for others with SCI. That is why he has formed the Mark Pollock Trust, which not only encourages SCI research but global collaboration. He and Simone are also directors on the Reeve Foundation board and have links with other SCI organizations in Europe. “We want to see this research enter health care systems and get to paralyzed people. That’s why I want to continue collaborating with UCLA and NeuroRecovery Technologies [see below], not only in America but also in Ireland. With the baseline data we have, it makes sense to me that if something starts to work, then you approach it like a training program. To build what we started in Los Angeles over a longer timeframe and include more people to see what the cumulative effect might be. The end goal is to move useful therapy from the laboratory to the people rapidly.”
On February 19, a consortium organized by the National Institute of Biomedical Imaging and Bioengineering, the newest NIH institute, held its second meeting with the goal of doing just that — fast tracking the research to move it to the public domain. According to Edgerton: “NIBIB is putting a high priority on trying to figure out the best ways to move the present technology to the SCI population as effectively and quickly as possible.” While the consortium’s main goal is to translate the latest research results into practical use, they are also proposing to organize specific trials on the use of epidural stimulation to improve bladder function.
Edgerton says they hope the epistim/bladder trial will serve as a way of not only addressing another important quality of life concern for people with SCI, but also function as a test of the consortium as a model of future effectiveness and collaboration.
Resources:
• Mark Pollock’s website: www.markpollock.com
• Mark Pollock Trust: www.markpollocktrust.org
• NeuroRecovery Technologies: Neurorecoverytechnologies.com
From Technology to Product
The medical device adapted for use in the Louisville epidural stimulation trials was an off-the-shelf implantable stimulator made by Medtronics for pain control. Now the race is on to bring an improved next-generation epidural stimulator, as well as a transcutaneous stimulator, to market. Nick Terrafranca, CEO of NeuroRecovery Technologies, says the need for more highly programmable electrical stimulation devices was the catalyst for the startup of the new company, whose scientific team, led by UCLA’s Reggie Edgerton, includes Yuri Gerisimenko, Roland Roy, Parag Gad and Wentai Liu, also of UCLA. Rounding out the team are Joel Burdick and Y.C. Tai of California Institute of Technology. The company hopes to have improved stimulation devices ready for widespread use within two to five years.
Support New MobilityWait! Before you wander off to other parts of the internet, please consider supporting New Mobility. For more than three decades, New Mobility has published groundbreaking content for active wheelchair users. We share practical advice from wheelchair users across the country, review life-changing technology and demand equity in healthcare, travel and all facets of life. But none of this is cheap, easy or profitable. Your support helps us give wheelchair users the resources to build a fulfilling life. |




Recent Comments
Kevin Hoy on TiLite Releases Its First Carbon Fiber Wheelchair
tuffy on NYDJ Launches Women’s Adaptive Jeans on QVC
Lisa Cooley on How to Fund an Expensive Adaptive Vehicle